
This toolkit was created to serve as a resource for state lawmakers, staff, patient advocates, media and others who want to better understand the pharmaceutical policy landscape — and act to improve it. It is grounded in free-market principles, centered on patients, and committed to protecting innovation as a vital public good.
(Editor’s note: The author would like to thank the policy team at the Goldwater Institute. Several of the recommendations described in this report were pioneered by the Institute.)
The breakthroughs we now take for granted — new cancer treatments, cures for once-fatal diseases, drugs that lead to longer and healthier lives — exist because the United States has long led the world in pharmaceutical innovation.
That leadership, however, is no longer guaranteed.
China is rapidly expanding its pharmaceutical sector, aiming to surpass the U.S. in drug development and biomanufacturing. At the same time, federal policies are increasingly undermining the ecosystem that made American innovation possible.
The price controls in the Biden administration’s Inflation Reduction Act are already discouraging investment in high-risk, high-reward research areas like rare diseases and oncology. While voluntary, manufacturers may offer Most Favored Nation pricing, which is based on foreign countries’ drug price rationing schemes, through the Medicaid program. And the looming reorganization of the FDA threatens to create additional bottlenecks and delays at a time when speed and certainty are urgently needed. These obstacles and uncertainty, taken together, could weaken U.S. leadership in drug innovation. That matters because it can slow new cures and push research, jobs, and investment overseas.
States matter even more in this new reality.
While Washington debates and delays, state lawmakers are positioned to protect patient access, encourage competition and foster an environment where medical innovation can thrive. States control major levers of pharmaceutical policy, including drug pricing regulations, pharmacy benefit manager practices, Medicaid purchasing, prescribing rules, and transparency requirements. The actions taken at the state level will play a critical role in determining whether future cures are developed here, somewhere else, or not at all.
This toolkit was created to serve as a resource for state lawmakers, staff, patient advocates, media and others who want to better understand the pharmaceutical policy landscape — and act to improve it. It is grounded in free-market principles, centered on patients, and committed to protecting innovation as a vital public good.
Inside are ideas that focus on reforms that would:
Expand competition to lower costs and empower patients.
Remove outdated regulatory barriers that restrict access and choice.
Strengthen transparency and accountability in the pharmaceutical supply chain.
Protect and accelerate innovation for future treatments and cures.
The future of medicine — and the ability for patients to benefit from it — depends on the decisions we make today. With the right reforms, states can lead the way, ensuring a health care system that delivers affordability, accessibility and innovation not just for today’s patients, but for generations to come.
America’s innovative pharmaceutical sector has delivered cures and breakthroughs once thought impossible. But the benefits of innovation must be balanced with policies that ensure patients can access treatments that improve and extend lives. Competition is the key.
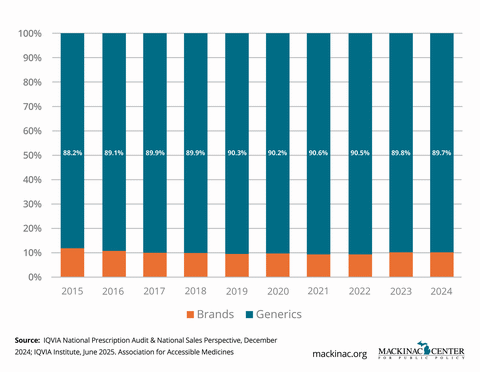
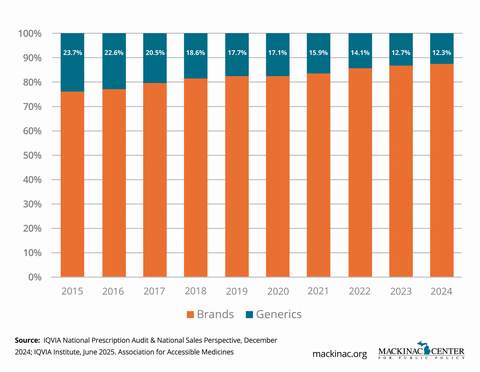
Generics and biosimilars prove this. These are drugs that are identical or near identical to patented and already approved drugs. When generics and biosimilars enter the market, drug prices drop dramatically, sometimes by as much as 85% — giving patients more choices and relief at the pharmacy counter.
Yet too often, competition is delayed or blocked. Brand-name manufacturers may use complex patent strategies, minor product changes ("product hopping"), and legal loopholes to extend patent monopolies far beyond the original innovation. In some cases, generic companies face years of expensive litigation just to bring lower-cost alternatives to the market. These delays keep prices artificially high and limit patient access to affordable medicines.
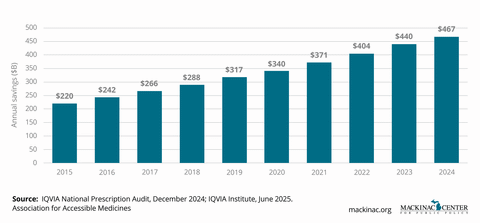
Generics and biosimilars save Americans hundreds of billions of dollars each year. The Association for Accessible Medicines estimated the figure to be $467 billion in 2024. In the oncology market alone, biosimilars are already projected to save billions of dollars over the next decade.
These savings are not hypothetical. They translate directly into better access and real-world benefits for patients managing chronic or life-threatening conditions.
Generics and biosimilars are proven success stories. When market barriers are removed, they unleash competitive forces that lower costs dramatically. Generic penetration rates typically exceed 90% within a few years of entry, leading to rapid price declines. Biosimilars, though newer to the U.S. market, are following a similar pattern in areas like oncology, immunology and endocrinology.
New competitors can create positive ripple effects across the health care system — encouraging brand-name manufacturers to offer patient assistance programs, investing in new innovations, or reducing prices voluntarily to maintain market share.
Despite these clear benefits, significant barriers remain. "Patent thickets" are dozens of overlapping patents on minor elements of a drug. They are strategically deployed to block competition. "Product hopping" tactics slightly tweak a formulation or dosage to reset patent protections, even when no meaningful clinical benefit exists. Meanwhile, biosimilars often face marketing challenges and a patchwork of state-level rules that limit pharmacist substitution and slow adoption even after FDA approval.
The result is a marketplace where patients wait years longer and pay significantly more than necessary for affordable alternatives.
Support competitive contracting: Encourage state Medicaid programs and taxpayer-financed public employee health plans to prioritize clinically appropriate generics and biosimilars in their formularies, which are the lists of prescription drugs covered by the health plan.
Ban product hopping: Enact laws that prevent patients from being switched to new formulations of brand drugs that are about to lose market exclusivity. This product hopping prevents patients from being switched to generic alternatives.
Promote pharmacist substitution: Allow pharmacists to automatically substitute FDA-approved generics and biosimilars, with reasonable notification requirements, without placing undue burdens on patients or providers.
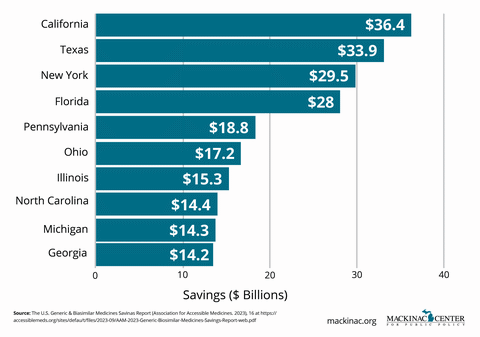
Michigan patients, consumers and taxpayers saved more than $14 billion in 2024 thanks to generics and biosimilars. These savings provide real-world benefits: every dollar saved represents the ability to provide more patient access and care using fewer resources and without compromising the quality of care. Expanding competition does not just lower costs — it empowers patients, strengthens access, and fuels a healthier, more innovative pharmaceutical marketplace.
New treatments and affordable alternatives can only transform lives if patients can access them. Yet too often, regulatory delays — at both the federal and state levels — keep promising and proven, cost-saving therapies out of reach for patients who need them.
Smart regulatory reforms can safely accelerate access, lower costs and expand choice. Conversely, outdated rules, duplicative licensing requirements and slow adoption policies stifle innovation and drive-up prices. States that modernize their regulatory frameworks can deliver faster, better and more affordable care to their citizens.
For example, even after FDA certification, some biosimilars and generics face years of restricted use due to slow formulary updates, cumbersome pharmacy laws and insurer inertia. While the FDA sped up approvals without sacrificing safety with programs like Real-Time Oncology Review and Project Orbis, post-approval hurdles still loom large. Patients and taxpayers continue to shoulder higher costs as a result.
Our regulatory system helps ensure the safety and effectiveness of medicines. Modern initiatives at the FDA demonstrate that careful streamlining is possible without compromising patient protection. When these efficiencies are embraced — not just federally, but at the state level, too — patients gain faster access to safe, lower-cost alternatives.
Successful examples include early state adoption of biosimilar substitution laws and Medicaid policies that actively incorporate newly approved generics into preferred drug lists. These states are seeing earlier savings and broader patient access as a result.
Despite these pockets of progress, inertia dominates many areas. Formularies are often updated only annually, meaning patients wait months for newly available lower-cost options. State pharmacy laws sometimes prevent pharmacists from substituting biosimilars for brand-name biologics even when federal law permits it. Licensing and substitution requirements vary widely between states, creating regulatory friction that discourages competition.
The patchwork system not only drives up costs but also reinforces inequities — where a patient’s access to affordable medicine can depend more on their zip code than on medical need.
Streamline licensing and substitution laws: Simplify pathways for pharmacists to substitute FDA-approved generics and biosimilars without unnecessary barriers.
Mandate fast formulary updates: Require state Medicaid programs and state employee health plans to evaluate and adopt newly approved generics and biosimilars within a set period (such as 90 days after FDA approval).
Prioritize accelerated approval products: Ensure public health programs consider FDA-approved products developed through accelerated pathways alongside traditional therapies.
Lowering costs and expanding choice is not just delivering more drugs — it is about smarter, faster access to safe, proven treatments when patients need them most.
Patents exist to motivate and reward innovation. They give companies the exclusive right to market a new drug for a period, allowing them to recoup research costs and invest in future breakthroughs. But today, some companies exploit loopholes in the system to block competition and extend monopolies far beyond the original brand exclusivity timeframe.
Strategies like "patent thickets," "evergreening," and "pay-for-delay" settlements can prevent lower-cost generics and biosimilars from reaching patients for years — even decades. These tactics protect profits at the expense of competition, undermining affordability, access and patient choice.
A 2022 analysis published by the Initiative for Medicines, Access, and Knowledge found that brand-name drug manufacturers often file dozens or even hundreds of patents on the same product, covering minor changes like dosage forms, delivery methods or packaging. This creates a "thicket" of legal hurdles that deters generic challengers. Meanwhile, settlements between brand-name and generic companies sometimes involve payments to delay entry, undermining competition and costing patients billions.
Intellectual property protection encourages investment and innovation. The promise of a time-limited monopoly motivates companies to take the risks necessary to develop breakthrough therapies. When patents are granted appropriately, they can drive medical progress that benefits patients worldwide.
When patent protections are abused, they become tools of market concentration and distortion rather than innovation. Patients and taxpayers end up paying monopoly prices long after a product's justified privilege of exclusivity ended. Competition is stalled, costs remain high and incentives to invest in new therapies diminish.
Patent abuse not only keeps prices high, it also hurts the credibility of the entire innovation system by fueling public skepticism about whether patents protect ideas or just profits.
Prohibit pay-for-delay agreements: Ban contracts in state antitrust law where brand-name companies pay generic competitors to delay market entry.
Support transparency: Require companies that market prescription drugs in the state to disclose the number of patents covering each product.
Challenge sham patents: Create incentives for state attorneys general or consumer protection divisions to challenge obvious or abusive patent thickets.
Pharmacy Benefit Managers, or PBMs, were created to negotiate lower drug prices for insurers, employers and public health care programs. In theory, they should save patients money by leveraging their bargaining power to drive down costs. But in practice, the largest PBMs have grown into vertically integrated giants that control much of the prescription drug marketplace — with mixed results for patients, employers and taxpayers.
According to the Federal Trade Commission, three companies — CVS Caremark, Express Scripts, and OptumRx — control nearly 80% of the prescription drug market. These entities are owned by or tightly affiliated with major insurers and pharmacy chains, which further concentrates their market power.
PBMs can and do negotiate significant discounts on behalf of smaller insurers, employers and consumers. Transparent, “pass-through” PBM models deliver savings by charging a flat administrative fee without profiting from “spread pricing” or hidden rebates. Discount programs like GoodRx, which rely on PBM networks, can provide crucial savings for uninsured and underinsured patients.
Done right, PBM negotiation can help lower prescription drug costs while maintaining access to necessary medications.
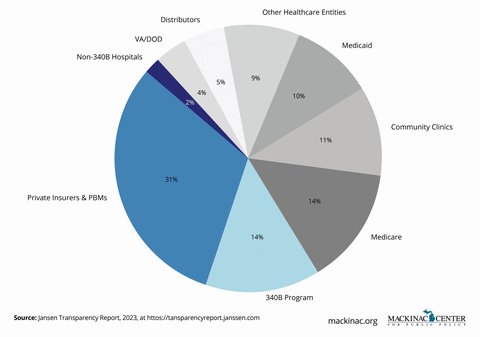
The dominant PBMs engage in practices that obscure true drug costs and distort patient choice. Spread pricing, for example, is the difference between what a PBM pays to a pharmacy for a drug and what it charges the insurance plan sponsor. The “spread” is the profit in these transactions. Spread pricing, opaque rebate structures, gag clauses and steering patients to owned pharmacies or formularies are common. These tactics inflate costs, limit access and shield pricing practices from scrutiny.
Recent rebate transparency reports show that a significant share of discounts negotiated by manufacturers is absorbed by PBMs and insurers — not passed on to patients. Employers and state health plans often sign contracts that permit these practices, fueling cost growth without realizing intended savings.
Ban spread pricing: Prohibit PBMs from engaging in spread pricing in state-regulated health plans, including Medicaid. Half of the states, including Michigan, limit or ban this practice across various markets.
Mandate rebate transparency: Require PBMs to disclose negotiated rebates and pass savings directly to health plans and consumers.
Promote cash discount options: Incentivize health plans to allow patients to choose the lower of the insurance copay price or the cash price at the pharmacy counter. Patients should be able to apply cash payments to their prescription drug deductible.
Strengthen oversight: Establish licensing and audit requirements for PBMs operating in the state.
Pharmacists are among the most accessible health care professionals in America. Yet in many states, outdated regulations limit what pharmacists are allowed to do — even when it could improve patient care, expand access and lower costs.
Expanding the scope of pharmacist practice can play a critical role in a more affordable, patient-centered health care system. Allowing pharmacists to substitute biosimilars, prescribe for minor conditions, and manage chronic therapies under collaborative agreements with physicians could dramatically enhance care, particularly in underserved areas.
Pharmacists already play an important role in medication management, patient counseling and vaccinations. During the COVID-19 pandemic, emergency measures allowing pharmacists to test and vaccinate showed that expanded roles can be implemented safely and successfully.
Increased pharmacist authority, particularly for biosimilar and generic substitution, improves competition by speeding patient access to lower-cost, FDA-approved therapies.
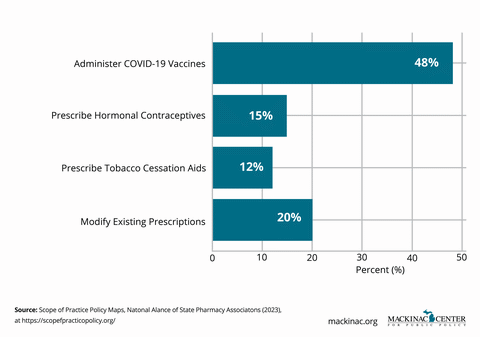
Many states still impose unnecessarily restrictive laws that limit pharmacist substitution, prescribing or collaborative care authority. These barriers raise costs and exacerbate access challenges, especially in rural and underserved communities.
Expand biosimilar substitution laws: Allow pharmacists to substitute FDA-approved interchangeable biosimilars unless explicitly prohibited by the prescriber.
Authorize pharmacist prescribing: Enable pharmacists to prescribe medications for routine, low-risk conditions under a state’s standing orders or collaborative agreements.
Support chronic disease management programs: Permit pharmacist to -diagnose and prescribe maintenance medications for chronic conditions like hypertension and diabetes.
Protect consumer choice: Prevent PBMs or insurers from “steering” patients to their pharmacies and enable consumers to choose their own pharmacy.
Streamline licensing: Harmonize licensing standards across states to promote workforce mobility through the adoption of universal license recognition and reciprocity agreements.
“Telepharmacy” — accessing a pharmacist remotely — and remote dispensing technologies are changing the way patients access medications, particularly in rural and underserved areas. These innovations allow patients to receive timely prescription services even when brick-and-mortar pharmacies are miles away.
By embracing telepharmacy, states can expand access to care, support medication adherence and promote competition.
Remote dispensing models allow licensed pharmacists to supervise pharmacy technicians via real-time, audio-visual links. Patients can speak directly to pharmacists, receive counseling, and then pick up prescriptions locally, without having to travel long distances.
In states that have adopted telepharmacy programs, early evidence shows improved medication adherence and high patient satisfaction. Remote dispensing sites can operate with lower overhead costs, sustaining pharmacy access in vulnerable communities.
Regulatory uncertainty and outdated pharmacy laws block the growth of remote dispensing in many states. Restrictive rules on pharmacist supervision, facility licensing, or patient verification requirements can make telepharmacy infeasible — even when it meets modern safety standards.
Modernize pharmacy laws: Clearly authorize full remote dispensing and telepharmacy operations. Twenty-eight states, including Michigan, have authorized some form of telepharmacy.
Expand access requirements: Allow telepharmacy models to operate anywhere in underserved areas and remove distance restrictions. Some states, including Michigan, require remote pharmacies to locate at least 10 miles away from existing, traditionally licensed pharmacies.
Ensure high-quality patient counseling: Require real-time pharmacist interaction for remote dispensing.
Promote competition: Remove protectionist rules that restrict telepharmacy licenses to existing brick-and-mortar chains.
Support rural health care networks: Integrate remote pharmacy access into rural health planning.
Telepharmacy isn’t just a convenience, it is a critical tool for ensuring access, lowering costs and strengthening health care delivery in every zip code.
Drug pricing in the United States is notoriously complex, and the confusion around what a drug costs often fuels frustration, distrust and poor policymaking. Too often, policymakers and the public focus on the list price of a drug — the price set by manufacturers — which almost no one pays.
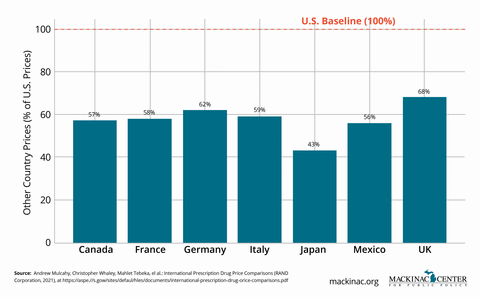
The reality is that the system is full of rebates, discounts and intermediary negotiations. Pharmacy Benefit Managers, insurers, wholesalers, pharmacies and government programs all negotiate and collect discounts at different stages of the supply chain. These behind-the-scenes discounts can be significant, sometimes exceeding 50% or more of the list price.
Yet because patients’ out-of-pocket costs are often tied to the inflated list price (rather than the discounted price), many people feel the full sting of high prices even though the system is supposedly offering deep discounts elsewhere.
The U.S. system does reward innovation and has enabled thousands of life-saving treatments to reach the market. Negotiation among private actors, particularly for generics and biosimilars, often achieves significant savings without government-set price controls. Rebates can and do lower net costs for insurers, government programs, and large employers.
Patients often do not benefit directly from negotiated savings. Because deductibles, co-insurance and out-of-pocket maximums are often calculated using list prices rather than net prices, patients pay more even if their insurer or pharmacy benefit manager is paying less. Moreover, lack of price transparency throughout the supply chain fuels inefficiency, distorts incentives and limits competition. And while generics often offer affordable options, U.S. brand drug price, as well some generic prices, can vary dramatically compared to other countries.
Take, for example, the generic for Singulair. It is used to manage asthma and allergies. The generic, Montelukast, has a list price of just under $100 for a 30-day supply. It can be found for under $10 through Amazon.com and GoodRx. Some U.S.-based mail order pharmacies sell a 30-day supply for under $1.
Expand access to generics and biosimilars: Ensure state programs aggressively adopt lower-cost alternatives as soon as they are available.
Ban list-price-based co-insurance: Mandate that patient cost-sharing in taxpayer-funded health programs be based on the net price after rebates, not the inflated list price.
Support real-time benefit tools: Require state health plans to implement electronic systems that show prescribers and patients the true cost of a drug at the point of prescribing.
Promote transparent formularies: Require public disclosure of formulary designs and rebate arrangements in taxpayer-funded health programs.
Understanding how drug pricing works is the first step toward building a system that is affordable, accountable, and fair.
It is often said that Americans pay more for prescription drugs than patients anywhere else in the world. That’s true — but the story is more complicated than it seems.
On an apples-to-apples basis, U.S. list prices for brand-name drugs are higher than in most developed countries. But U.S. patients also have faster access to new therapies, more treatment choices and greater investment in life sciences innovation. Other countries achieve lower prices largely by restricting access to drugs, delaying approvals and limiting available options for patients.
In many cases, American patients get access to new treatments years before those in other countries — if they get access to them at all.
The U.S. remains the global leader in pharmaceutical innovation, responsible for many new drug discoveries. American patients benefit from earlier access to the latest therapies and broader treatment options. In some cases, this early access can extend someone’s life significantly. Additionally, generic competition tends to occur much faster in the U.S., leading to steep price declines when exclusivity periods expire.
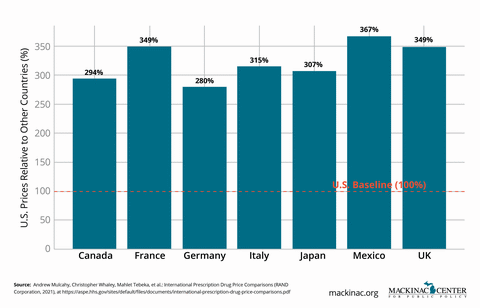
High list prices place significant financial strain on uninsured patients, those with high-deductible health plans and taxpayers. Rising out-of-pocket costs exacerbate affordability challenges. Moreover, rebates negotiated behind the scenes by PBMs and insurers are often not passed directly to patients at the pharmacy counter.
Expand access to generics and biosimilars: Ensure state programs aggressively adopt lower-cost alternatives as soon as they are available.
Ban list-price-based co-insurance: Mandate that patient cost-sharing in taxpayer-funded health programs be based on the net price after rebates, not the inflated list price.
Support real-time benefit tools: Require state health plans to implement electronic systems that show prescribers and patients the true cost of a drug at the point of prescribing.
Promote transparent formularies: Require public disclosure of formulary designs and rebate arrangements in taxpayer-funded or -supported health programs.
The U.S. health care model is far from perfect, but it delivers faster cures and more choices. Smart reforms can protect those features while making medicines more affordable for everyone.
For many patients, the shock of high drug prices is felt at the pharmacy counter. Rising drug deductibles, coinsurance tied to inflated list prices and increasingly narrow formularies mean that patients often pay more out-of-pocket even when insurers and pharmacy benefit managers negotiate large discounts behind the scenes.
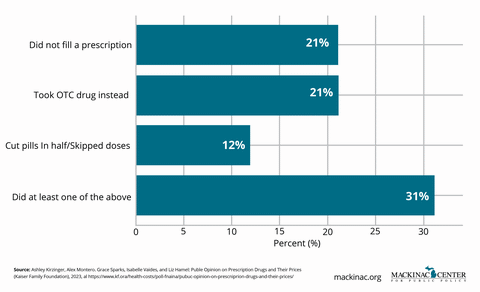
Sticker shock does not just cause frustration. It can lead patients to skip doses, delay filling prescriptions or abandon treatment altogether. This can ultimately lead to worse health outcomes and higher long-term health care costs.
Efforts to cap out-of-pocket costs, particularly for insulin and chronic disease medications, are gaining momentum across the country. Some states have already implemented maximum monthly co-pays for certain categories of drugs in state-regulated plans.
When patients can predict and afford their medication costs, they are more likely to continue their drug prescription, which means health outcomes improve, and overall system costs decline.
In many insurance plans, patients still pay coinsurance based on a drug’s list price rather than the discounted rate negotiated by the insurer or PBM. High deductibles often force patients to pay the inflated price until the deductible is met. Some plans also aggressively tier specialty drugs, shifting more costs onto patients, regardless of clinical need.
Sticker shock policies focused solely on capping prices without addressing underlying pricing dynamics risk adding short-term relief while masking longer-term structural issues.
Expand access to co-pay assistance programs: Prohibit insurers and PBMs from banning co-pay coupons or patient assistance programs.
Mandate real-time cost information: Ensure patients receive real-time pharmacy cost information when being prescribed medications, including the pharmacy retail price when it is lower than the required co-pay
Predictable, affordable access to medications is fundamental to ensuring that the treatments we develop reach the patients who need them.
State Medicaid programs are among the largest purchasers of prescription drugs. With that buying power comes an enormous opportunity to drive better pricing, competition and innovation — if states are willing to rethink traditional purchasing models.
Medicaid rebates and supplemental rebate negotiations already save states billions of dollars. But smarter, more flexible purchasing strategies can generate even greater savings and access improvements for low-income patients.
Medicaid already uses mandatory federal rebates to lower drug costs. Many states also negotiate additional supplemental rebates or participate in multi-state purchasing pools to enhance bargaining power. Value-based contracts, where payment is tied to patient outcomes, are beginning to emerge as new tools for smarter Medicaid drug purchasing. With the right policies, Medicaid can drive lower costs while encouraging better care and innovation.
Traditional Medicaid pharmacy benefit structures often reward volume rather than value. Bureaucratic hurdles can slow the adoption of new, cost-saving therapies. In some cases, spread pricing and opaque PBM practices in Medicaid Managed Care Organizations have undermined transparency and savings.
Federal rebate rules can also disincentivize aggressive state-level negotiation, locking states into complex administrative processes that may not optimize outcomes.
Ban spread pricing in Medicaid: Require pass-through PBM models where all discounts are passed directly to taxpayer-funded health programs, including the Medicaid program.
Encourage the use of online and mail-order pharmacy vendors: These pharmacies both encourage adherence to medications, often automating delivery and providing a 90-day supply with lower administrative costs to the health program.
Join or expand purchasing pools: Maximize negotiating leverage by partnering with other states where appropriate.
Demand full transparency: Require PBMs and MCOs to disclose pricing practices, rebate pass-through rates, and conflicts of interest to government payers.
Smart Medicaid purchasing means building a system that delivers better care at a sustainable cost.
In today's health care system, patients often feel trapped by restrictive prescription transfer rules that tie them to specific pharmacies, health plans or provider networks. Inflexible systems make it harder for patients to choose lower-cost options, access better service or maintain consistent care.
Prescription transfer reform is a low-cost, high-impact policy change that can give patients more control, improve affordability and increase competition among pharmacies.
Allowing patients to easily transfer prescriptions between pharmacies promotes competition based on service, convenience and price. Patients can shop for lower-cost options, seek better customer service or align pharmacy choices with their broader health care needs.
States that have adopted streamlined prescription transfer policies during emergencies, such as during fires and hurricanes, achieve better medication adherence. These transfers can be completed automatically without requiring patients to stand in line at the pharmacy or make multiple phone calls.
Patients, who are supposed to have access to and own their medical records, should be able to seamlessly use their own prescription history information to transfer their prescriptions to lower-cost or more convenient pharmacies. This can be done through APIs on their phone to search for the best prescription prices or the easiest delivery method.
Low-income patients, elderly individuals and rural residents often face the most difficulty in attempting to transfer their prescriptions to different pharmacies. This can require multiple phone calls or long waits at pharmacies.
Guarantee freedom of pharmacy choice: Protect patients' right to transfer prescriptions to any willing, licensed pharmacy. State lawmakers should adopt a Pharmacy Transfer Act, which applies to non-scheduled drugs, allowing for the easy transfer of prescriptions across pharmacies.
Ban anti-competitive steering: Prohibit PBMs and insurers from mandating use of owned or preferred pharmacies without patient opt-out rights.
Streamline electronic transfers: Mandate that prescription transfer systems between pharmacies operate electronically, reducing patient burden.
Empowering patients to seamlessly shop their prescriptions is one of the simplest and most powerful ways to promote affordability and quality in health care.
The way patients access medications is changing. From online pharmacies and mail-order fulfillment to manufacturer-run programs and telepharmacy partnerships, patients can access a growing share of prescriptions directly. In many cases, this approach is more affordable and transparent than traditional practices.
This “direct-to-patient” movement reduces reliance on costly middlemen that have long dominated the pharmaceutical marketplace. By connecting patients directly with licensed pharmacies or manufacturers, these models can deliver medications directly to someone’s doorstep. This will expand access in underserved areas and introduce new competitive pressures to put downward pressure one costs.
Across the country, innovators are already proving the potential of direct-to-patient models. Platforms such as Cost Plus Drugs and Amazon Pharmacy, along with smaller telepharmacy networks, are demonstrating that safe, affordable drug delivery can happen at scale. Patients can compare prices, receive prescriptions by mail or courier and connect with pharmacists through video consultations.
These models are especially valuable for rural residents and patients with limited mobility, for whom a trip to a physical pharmacy can be a barrier to care. Transparency is another hallmark: patients can see what they are paying for, without hidden markups or opaque rebate-driven pricing.
Direct-to-patient models can improve convenience and medication adherence, particularly for patients managing chronic conditions. Home delivery, 90-day fills and reliable refill scheduling can reduce missed doses and eliminate unnecessary delays.
A direct-to-patient method also strengthens competition. When patients can compare prices and choose delivery options that work for them, the pharmacies have to compete on service, reliability and cost.
Just as important, direct-to-patient models can expand access in communities where pharmacy closures, workforce shortages and long distances make routine prescription pickups difficult. Telepharmacy and remote pharmacist counseling can extend professional support to patients who otherwise face delays or go without it.
State policy has not kept pace with technological progress. Many pharmacy laws were written for a pre-digital era and assume that every prescription must be filled in person under a pharmacist’s physical supervision. Some states limit remote verification, restrict telepharmacy operations, or create unnecessary barriers for nonresident pharmacies — even when safety and accountability safeguards exist.
Pharmacy Benefit Managers and insurers often reinforce these barriers by steering patients toward preferred, vertically integrated pharmacy networks, often restricting access to lower-cost direct-to-patient options. And in many states, Medicaid and public employee health plans still lack clear authority, contracting flexibility or modern infrastructure to partner with transparent direct-to-patient vendors, leaving significant savings and access gains untapped.
The result is a system that keeps patients tethered to intermediaries, obscures true prices and restricts choice, leading to higher out-of-pocket costs and delayed access to medications that could be delivered safely within days.
Modernize pharmacy and telepharmacy laws: Update pharmacy practice acts to clearly authorize direct-to-patient dispensing models, including remote pharmacist oversight, remote verification where appropriate and telepharmacy counseling. This can be achieve all while maintaining clear accountability, documentation and patient safety standards.
Protect patient choice and ban anti-competitive steering: Prohibit PBMs and insurers from forcing patients into preferred or vertically integrated pharmacies when a willing, licensed direct-to-patient option is available.
Integrate direct-to-patient models into state programs: Allow Medicaid and public employee health plans to pilot or contract with direct-to-patient vendors for maintenance medications and other appropriate therapies. This is especially important where home delivery can improve medication adherence, reduce administrative costs and expand access in rural or underserved areas.
Direct-to-patient care represents the next step in health care modernization — putting patients, not intermediaries, at the center of the system. By updating outdated laws and embracing responsible innovation, states can make medicine more affordable, accessible and transparent for every resident no matter their ZIP code.
Transparency is essential for accountability in any marketplace, and the pharmaceutical sector is no exception. Yet pricing in the drug supply chain remains one of the least transparent sectors in health care.
Manufacturers set list prices, but few know how much PBMs, insurers, wholesalers or pharmacies are actually paying — or profiting — at each step. Patients often do not know whether the price they pay reflects the true market value of the drug, an inflated intermediary markup, or a negotiated rebate that benefits someone else.
Shining a light on the supply chain can help drive down costs, empower patients and improve trust in the health care system.
Transparency initiatives in other areas of health care — such as hospital price disclosures and insurer cost-sharing calculators — are beginning to show promise. Empowered consumers make smarter choices.
Without transparency, it is difficult to identify where price inflation occurs or to target reforms effectively. Policymakers end up making sweeping proposals — like price controls — that harm innovation rather than solving the underlying problem. Patients and taxpayers lose trust in a system they view as opaque and unfair.
Promote transparency in PBM contracting: Require disclosure of rebates, fees, and pricing structures in contracts involving state health care programs.
Develop public dashboards: Create simple, publicly accessible dashboards showing real drug costs for major state health programs.
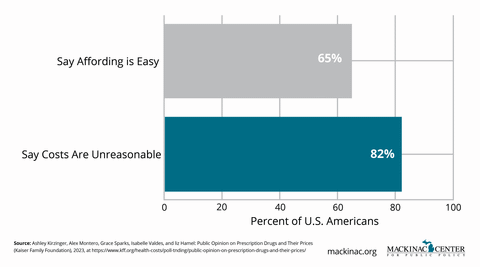
Transparency is not a silver bullet—but without it, meaningful reform is impossible.
Patients often assume that the high cost of a prescription drug is due to the manufacturer’s price. Multiple players — PBMs, insurers, wholesalers and pharmacies — each take a cut of the price before it reaches the patient.
Lack of transparency across the supply chain obscures true drug costs, creates perverse incentives and drives up prices unnecessarily. If states want to meaningfully address drug affordability, they must shine a light on the full journey a drug takes from factory to pharmacy counter.
Greater transparency across the health care sector is beginning to show real promise. Hospital price transparency rules and insurer cost calculators help patients make more informed choices. Transparency initiatives for the pharmaceutical supply chain could have similar benefits, forcing middlemen to justify pricing practices and helping policymakers craft more effective reforms.
Some states have already adopted laws requiring PBM rebate disclosures, while others are moving toward comprehensive supply chain reporting.
Patients still have little insight into who profits from each step of the drug distribution process. PBMs often pocket substantial rebates and fees without passing savings to patients. Wholesalers and pharmacies may add opaque markups. Insurers design formularies that maximize their own rebates rather than optimizing patient access.
Without clear, public reporting these hidden costs fester and drive misinformed policies that target manufacturers alone rather than addressing the entire system.
Require full, supply-chain reporting: Mandate disclosure of pricing, rebates and fees across PBMs, wholesalers, pharmacies and insurers for state-regulated plans.
Promote pass-through models: For taxpayer-funded health plans, require that PBMs and supply-chain structures pass negotiated discounts and savings directly to taxpayers.
Use data to inform reforms: Base affordability initiatives, such as price control proposals and prescription drug affordability boards, on data across the supply chain, not assumptions about one player.
Transparency empowers smarter choices, better reforms and more accountable systems that work for patients.
The 340B Drug Pricing Program was created with a simple, powerful goal: to help health care providers that serve low-income populations stretch their scarce resources. Under 340B, eligible hospitals and clinics can purchase outpatient drugs at steep discounts, then use the savings to fund uncompensated care.
Unfortunately, over time the program drifted from its original intent. Today, 340B has become a sprawling, loosely regulated system that too often benefits large health systems rather than the underserved patients it was designed to help.
When administered properly, 340B discounts help rural hospitals, community health centers and other safety-net providers expand services, lower drug costs for patients and strengthen access to care. Especially in underserved communities, 340B savings can mean the difference between a clinic staying open or closing its doors.
A couple of states have taken important steps to promote 340B program integrity through transparency requirements and targeted audits without undermining the program's core mission.
Program oversight at the federal level is minimal. There are no requirements to track how 340B revenues are used — or even to ensure that savings directly benefit low-income patients. Some large hospital systems exploit the program by contracting with for-profit retail pharmacies, generating substantial profits while providing little or no charity care.
Without stronger transparency, 340B risks undermining public trust and losing political support.
Mandate public reporting: Require 340B entities to disclose how savings are used to expand patient care services.
Prioritize patient benefit: Ensure that discounted drugs acquired through 340B are passed on to vulnerable patients.
Conduct regular audits: Authorize state health departments or auditors to review and publicly report 340B practices to ensure compliance with program intent.
Restoring the 340B program to its original mission will ensure that safety-net providers — not hospital profit centers — are the ones delivering real benefits to the patients who need them most.
Public opinion polls show that roughly three-quarters of Americans want stronger action to limit drug prices. That may be a driving factor explaining why Prescription Drug Affordability Boards are a growing policy tool that states are using to try to control rising drug costs. These boards are typically empowered to review high-cost drugs and impose upper payment limits for certain payers.
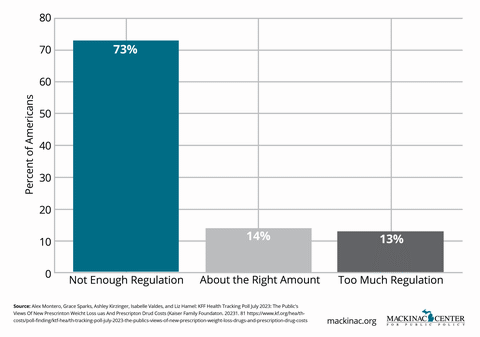
While well-intentioned, prescription drug affordability boards pose significant risks to innovation, access and patient care. They could slow the development of future treatments while doing little to address the true drivers of health care spending.
The motivation behind these drug boards — to increase accountability and affordability in drug pricing — is understandable. Patients facing high out-of-pocket costs need relief, and policymakers should seek to understand pricing structures.
Prescription drug boards also have the potential to collect and publish valuable data on drug pricing trends, providing lawmakers and the public with a better understanding of cost dynamics.
Prescription drug affordability boards often focus narrowly on drug prices in isolation, ignoring the complex interplay of PBMs, insurers, rebates and the broader health care system. Capping drug prices without addressing systemic inefficiencies risks unintended consequences, such as reduced access to innovative therapies.
These drugs boards also introduce significant uncertainty for manufacturers, which can chill investment in high-risk research areas like rare diseases and oncology. Programs that appear to cap prices may also create secondary rationing effects, forcing providers to navigate cumbersome reimbursement processes for patients needing targeted therapies.
States that consider prescription drug affordability boards must understand that to “do something” about drug costs may ultimately do more harm than good.
Mandate holistic analysis: Resist efforts to establish a prescription drug affordability board. Instead, state lawmakers should analyze and assess the full drug supply chain — including PBM practices, insurer negotiations and hospital markups.
Protect breakthrough therapies: Exempt innovative treatments for rare diseases, oncology and other high-risk areas from price regulation.
Prioritize patient access and cost savings: For states that do pursue a drug board, safeguard against access restrictions, ensure that its recommendations do not delay or limit treatments. States should make sure that the savings are passed directly to patients and taxpayers.
Use drug boards for data, not rationing: Focus the activity of prescription drug affordability boards on gathering and publishing pricing information rather than imposing rigid controls.
Affordability is vital — but rationing innovation and access is not the answer. States can seek transparency without sacrificing patient progress.
Medical science is entering a golden age. From gene therapies that can cure inherited diseases to personalized cancer vaccines and AI-designed drugs, the future of medicine promises transformative breakthroughs for patients who once had few or no options.
But as treatments become more sophisticated — and sometimes more expensive — states must prepare new frameworks for how health care systems, insurers and public programs will evaluate and pay for these innovations. Without thoughtful action, tomorrow’s cures could remain out of reach for the very people who need them most.
Breakthrough treatments deliver unprecedented results. Gene therapies for conditions like spinal muscular atrophy and beta thalassemia offer the potential for one-time, curative interventions. Immunotherapies are extending survival rates for cancers once considered terminal. New rare disease therapies are emerging at a faster pace than ever before.
Over the long term, many of these therapies can lower health care costs by reducing hospitalizations, surgeries and lifetime treatment expenses. Investing upfront in curative treatments can mean major savings for health care systems and a higher quality of life for patients.
Innovative payment models — such as installment plans, outcomes-based contracts and reinsurance mechanisms — are beginning to show promise in making high-cost therapies more affordable for both public and private payers.
Traditional insurance and Medicaid structures were designed for chronic management of disease, not single-administration cures. High upfront costs, even if cost-effective over time, pose major budget challenges for state Medicaid programs, employer plans, and uninsured patients.
Coverage decisions can be slow and inconsistent. Patients may face delays, denials, or narrow eligibility criteria that limit access to life-changing therapies. Without updated payment systems and smarter coverage policies, the promise of these breakthroughs could be squandered.
Fear of costs, if unaddressed, may also drive political and regulatory proposals that restrict innovation and ration future access.
Promote outcomes-based payment models: Encourage health plans and Medicaid to adopt payment structures that link costs to actual patient outcomes.
Support reinsurance pools for high-cost treatments: Develop risk-sharing mechanisms that spread costs for expensive therapies across multiple years.
Mandate timely coverage reviews: Require state health plans and Medicaid to review and cover FDA-approved breakthrough therapies within a reasonable timeframe.
Protect patient access: Enact laws that prevent exclusions of innovative treatments based solely on high upfront costs without comprehensive review of overall costs and potential savings in areas such as hospitalizations, disease burden and more.
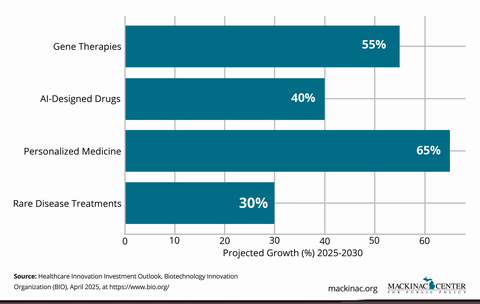
Science is moving fast. With smart, forward-looking policies, states can ensure that tomorrow’s medical breakthroughs are delivered to the patients who need them. Policymakers have the opportunity today to deliver the breakthroughs patients expect tomorrow.
The federal Right to Try Act, passed in 2018, was a landmark law that gave terminally ill patients access to investigational treatments outside of traditional clinical trials, without having to beg the federal government for permission to do so. But as medicine advances, a new frontier is emerging: individualized therapies, such as customized cancer vaccines, gene editing and personalized cell therapies.
Today’s laws were not designed for a future where a treatment might be designed for a single patient. States could lead by modernizing Right to Try policies to accommodate individualized treatments safely and ethically. Fifteen states have already begun expanding Right to Try laws to include the individualized treatment pathway.
Individualized treatments offer enormous hope for patients with rare, aggressive or treatment-resistant conditions. For some, accessing a cutting-edge, individualized therapy may be their only chance at survival.
Extending Right to Try frameworks to include individualized treatments can encourage responsible innovation, empower patients and extend lives.
Current Right to Try laws generally cover drugs or biologics that have completed Phase I trials. However, many personalized therapies are custom-made and do not fit traditional clinical trial models. Bureaucratic red tape, regulatory uncertainty and liability fears prevent broader use.
Without modernization, patients could be blocked from accessing life-extending personalized treatments simply because old laws do not recognize new realities.
Expand Right to Try definitions: Update statutes to clearly include individualized cell, gene, and molecular therapies.
Provide legal protections: Shield providers and manufacturers from liability when offering access under ethical and evidence-based standards.
Respect patient autonomy: Ensure that patients with life-threatening conditions have a clear, fair and expedited pathway to explore all treatment options. This includes, but is not limited to, gene and cellular therapies that are individualized to the specific patient.
Medicine is becoming personal. States can lead by making compassion, safety, and innovation the cornerstones of access.
As states search for ways to manage health care costs, some policymakers are considering using a metric known as quality-adjusted life years to set drug coverage and reimbursement decisions. This statistic attempts to measure the value of a treatment by estimating the additional years of life it provides, adjusted by the patient’s current quality of life. In theory, this can help prioritize resources. In practice, however, rationing based on quality-adjusted life years discriminates against people with disabilities, chronic illnesses and older populations.
Relying on quality-adjusted life years as the foundation for drug coverage can undermine patient access to critical therapies, limit choice and worsen health disparities. Worse still, it places bureaucratic judgments ahead of clinical expertise and patient needs.
Cost-effectiveness analyses can be helpful tools when used properly. Evaluating how much benefit a treatment provides relative to its cost encourages smarter spending and can help steer limited public resources toward high-impact care. States can use this value-based framework to better understand the economic implications of health care decisions without resorting to rigid formulas that devalue human life.
When cost data is used as one of many factors — rather than as the sole determiner — policymakers can achieve better outcomes without harming vulnerable patients.
The quality-adjusted life year framework assigns lower value to treatments for people with disabilities, the elderly and those with chronic conditions. Under these calculations, a year of life for a healthy young person is worth more than a year of life for someone living with a disability or serious illness. This approach leads to systemic discrimination against the very populations that often need health care advances the most.
Medicare explicitly bans the use of this measure for coverage decisions. Yet quality-adjust life year calculations are increasingly proposed for state Medicaid programs, Prescription Drug Affordability Boards and other pricing bodies.
Prohibit rationing based on quality-adjusted life years: Ban the use of quality-adjusted life years or similar metrics in state health care decision-making processes.
Encourage patient-centered metrics: Encourage the use of decision-making tools that measure value by patient outcomes, recognizing that people differ in their conditions, circumstances and priorities.
Demand transparency: Require any contractor or advisory board providing value assessments to disclose methodology and account for nondiscrimination protections.
Patients are not mathematical formulas. States can and must reject frameworks that reduce human lives to numbers and ensure that health care decisions reflect both medical science and fundamental human dignity.
Personalized medicine is here. Advances in genomics, biomarkers and molecular diagnostics are making it possible to tailor treatments to each patient’s unique biology. Instead of a one-size-fits-all approach, personalized medicine offers therapies that are safer, more effective, and, ultimately, more cost-efficient.
States that adapt to the personalized medicine revolution will not only improve health care outcomes but will also position themselves at the forefront of next-generation health care delivery.
Personalized medicine offers transformative benefits for patients and health systems. In oncology, genomic profiling allows for therapies targeted to specific mutations, dramatically improving survival rates. In cardiology, genetic testing can help tailor medications to minimize side effects and optimize dosing.
Early diagnosis through precision diagnostics can enable interventions that prevent disease progression, reducing long-term health care costs. Personalized approaches are showing promise in managing rare diseases and autoimmune conditions where traditional treatments often fall short, if they exist at all.
State policies, insurance frameworks for state self-insured plans, and Medicaid coverage rules often lag scientific advances. Many essential diagnostic tests are not covered by insurance, forcing patients to pay out-of-pocket or forego care. The lack of next-generation testing can lead patients on long diagnosis journeys that delay care and result in worse patient outcomes. Public health programs may resist integrating new personalized therapies into formularies due to outdated cost-effectiveness models.
There are also persistent disparities in access to genomic testing based on geography, income and race — risking a future where the benefits of personalized medicine accrue only to the privileged few.
Expand coverage for rapid whole-genome sequencing: Ensure that Medicaid and state-regulated plans cover clinically validated diagnostic tests for patients facing critical illnesses. To date, 17 Medicaid programs have expanded rapid whole-genome sequencing for critically ill infants.
Update medical practice regulations: Modernize standards of care to incorporate personalized and precision medicine where appropriate. For example, lawmakers can direct their State Medical Boards to promulgate policy to ensure that rapid genomic sequencing is a clinically appropriate diagnostic tool when ordered, consented, interpreted and followed up according to accepted professional guidance This could especially benefit care for critically ill infants and children with a suspected genetic diagnosis.
The future of health care is personal. States that prepare now will not only improve individual lives but also lead a national transformation toward better, more efficient and more equitable care across all areas of their state.
Discovering a cure is only half the battle. Ensuring patients can access it is just as critical. Without thoughtful access policies, breakthrough therapies remain out of reach for the people who need them most.
High costs, restrictive insurance coverage, step therapy requirements and bureaucratic delays are among the biggest barriers preventing patients from benefiting from cutting-edge treatments. States have a pivotal role to play in protecting access, especially for patients battling serious, rare or life-threatening diseases.
Recent years have seen an explosion of innovative treatments — from gene therapies for rare genetic disorders to breakthrough immunotherapies for cancer. When patients can access these therapies early, survival rates improve, quality of life increases and long-term health care costs can decline significantly.
Several states have begun adopting policies to streamline access, such as limits on step therapy protocols and expedited review processes for Medicaid coverage of new treatments.
Many patients must endure "fail-first" requirements, where insurers or Medicaid plans force them to try cheaper, less effective treatments that often fail before approving breakthrough therapies. Prior authorization processes can be opaque, slow and burdensome.
For rare disease patients, delays can be devastating. Many breakthrough therapies are time-sensitive, and bureaucratic barriers can mean the difference between life and death.
Limit step therapy requirements: Allow exemptions for patients when evidence supports direct access to FDA-approved breakthrough therapies.
Mandate rapid coverage decisions: Require Medicaid and state employee health plans to evaluate and cover new breakthrough therapies within 60 days of FDA approval.
Streamline prior authorization: Simplify prior authorization for innovative therapies and enforce timely response standards.
Protect access for rare disease patients: Ensure Medicaid programs have specialized coverage determination review pathways for rare or ultra-rare conditions.
Innovation cannot help patients if it stays locked behind regulatory barriers. Access must be part of the innovation conversation from the beginning.
Scientific knowledge evolves faster than government regulations. Physicians often rely on the latest studies, clinical experience and real-world evidence — not just FDA labeling — to treat their patients. Yet current federal rules restrict how manufacturers and researchers can share information about off-label uses of approved drugs.
Restricting this flow of information does not just limit free speech — it impedes patient care and slows innovation.
Doctors have always had the legal and ethical right to prescribe drugs off label based on their medical judgment. In many cases, off-label uses represent important advances in patient care, such as using an immunotherapy drug for a cancer patient for treatment in another part of the body than where the initial cancer appeared.
Allowing communication about off-label uses can help health care providers stay informed, personalize treatment decisions and improve patient outcomes.
Some court decisions have affirmed that scientific speech deserves First Amendment protection, but federal and state policies still often lag.
Manufacturers remain heavily restricted by the FDA in how they can share scientific information about off-label uses, even when the information is sound and medically promising. Fear of regulatory penalties discourages communication between researchers, companies and providers.
This chilling effect slows the adoption of important new evidence, especially in rural areas, and hinders the real-world optimization of approved therapies.
Affirm free scientific speech: Enact laws protecting the right for anyone to share accurate information about off-label uses.
When truth is censored, innovation slows and patients lose out on potential treatments. Protecting scientific communication is fundamental to advancing medicine.
Managing health care costs and promoting innovation are not mutually exclusive goals. In fact, protecting medical innovation is essential for building a system where better treatments drive affordability, competition and improved outcomes over the long term.
States must resist the false choice between affordability and innovation — and instead pursue policies that nurture both.
Strong intellectual property protections, robust competition frameworks and targeted affordability reforms have historically allowed the U.S. to remain the world leader in pharmaceutical innovation while introducing thousands of affordable generic and biosimilar alternatives.
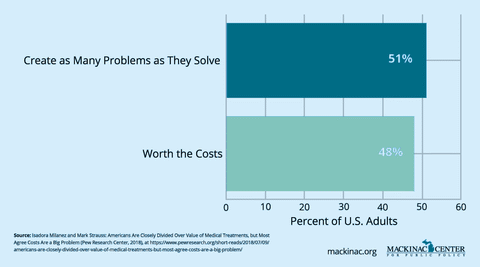
Market-based approaches — like paying for drugs based on how well they work, designing insurance benefits to steer patients toward high-value care and streamlining outdated approval and coverage processes — can lower costs while still rewarding innovation.
Blunt instruments like price controls and caps on payments or reimbursements risk investment in the very cures patients need. Policies that attack the drug development process without recognizing the high failure rate and risk involved may cause long-term harm by reducing the pipeline of future therapies.
Overregulation and uncertainty drive up risk and cost — ultimately harming patients by delaying or denying access to new treatments.
Support value-based pricing models: Encourage taxpayer-funded health plans that link reimbursements to outcomes rather than blunt price ceilings.
Promote transparent competition: Support faster generic and biosimilar adoption without punishing innovation.
Innovation and affordability are two sides of the same coin. When done right, protecting one strengthens the other — and both ultimately serve the patient.
The United States’ leadership in pharmaceutical innovation has produced life-saving breakthroughs, but that leadership is not guaranteed. At the same time, affordability and access challenges are real — often driven by delayed competition, outdated state-level rules and a supply chain that can obscure true costs and misalign incentives. The path forward is not to ration innovation, but to modernize policies so that patients benefit from both faster medical progress and more competitive pricing.
This toolkit outlines practical steps state lawmakers can take to strengthen competition and reduce costs without sacrificing patient outcomes. These include accelerating generic and biosimilar uptake, removing unnecessary barriers to prescribing and telepharmacy, curbing anti-competitive tactics that delay market entry, and bringing transparency and accountability to the whole supply chain. States can also improve purchasing and benefit design in Medicaid and other taxpayer-supported plans — ensuring faster formulary updates, eliminating list-price-based cost sharing and using data-driven approaches that target the real sources of price inflation.
Finally, as the next wave of personalized and potentially curative therapies arrives, states should modify their laws and policies to match the moment — timely coverage decisions, patient-centered value measures and payment models that can handle high upfront costs while rewarding real-world outcomes. With clear rules, open markets and accountable intermediaries, states can help ensure that tomorrow’s cures are discovered, developed and delivered to patients.